|
Its alloys are used in jewelry and for coins. Also this element is used in the manufacture of water pipes. 2 of them are stable, 1 of which is naturally occurring.Ĭopper is most often used as an electrical conductor. When in moist conditions, a greenish layer forms on the outside of it. Copper atoms have 29 electrons and 29 protons with 34 neutrons in the most abundant isotope. Copper radionuclides offer a varying range of half-lives and decay. Copper is a malleable, ductile and an excellent conductor of heat and electricity. Natural copper comprises two stable isotopes, 63 Cu (69.17) and 65 Cu (30.83), and 27 known radioisotopes, five of them are particularly interesting for molecular imaging applications (60 Cu, 61 Cu, 62 Cu, and 64 Cu), and in vivo targeted radiation therapy (64 Cu and 67 Cu). Mass number (number of nucleons) (29 protons and 34 neutrons) 63 29 Cu 65 29. This element was extracted and used for thousands of years. The isotopic notations for the two isotopes of copper are written as follows. Please contact us via request a Copper-63 quote to order Copper-63 isotope, to get Copper-63 price and to buy Copper-63 isotope.Ĭopper is a red-brown transition element. 
Copper-63 isotope is used for as a reference in elemental analysis through secondary ion mass spectrometry Ĭopper-63 isotope is available to order from in Copper-63 metal (Cu) chemical form and in Copper-63 oxide (CuO) chemical form. How many neutrons are present in each of these isotopes Why are they all considered aluminum atoms, even though they differ greatly in mass Write the atomic symbol for each isotope.Copper-63 isotope is used for studies of energy emissions in nuclear reactors.Copper-63 isotope is used for production of background-free tritium targets.One has 29 protons and 34 neutrons in the nucleus the other has 29 protons and 36 neutrons (Figure 4). Further data for naturally occuring isotopes of copper are listed above. But they also contain uncharged particles called neutrons. Copper-63 isotope is used for studies of impact of radiation on irregularities in material structures using methods of nuclear gamma resonance (NGR) and electron paramagnetic resonance (EPR) Do all copper atoms have the same number of neutrons All copper atoms have atomic number 29: all their nuclei contain 29 protons.Copper-63 isotope is used for studies of high-temperature superconductivity. 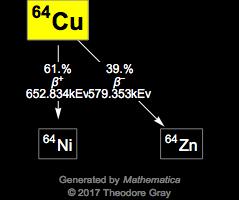
Copper-63 isotope is used for neutron spectrum measurement.Copper-63 isotope is used for Copper-62 (Cu-62 isotope, 62Cu isotope) positron-emitting radionuclide (radioisotope) production (can be used in life science for healthcare and medical applications and pharmaceuticals industries).Copper-63 isotope is used for Copper-64 (Cu-64 isotope, 64Cu isotope) radionuclide (radioisotope) production (can be used in life science for healthcare and medical applications and pharmaceuticals industries).Copper-63 isotope is used for Zinc-62 (Zn-62 isotope, 62Zn isotope) radionuclide (radioisotope) production (can be used in life science for healthcare and medical applications and pharmaceuticals industries).Copper-63 isotope ( Cu-63 isotope, 63Cu isotope)
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
